Periodic Table Families Periodic Table That Shows Solids Liquids and Gases
Chapter 2. Atoms, Molecules, and Ions
ii.5 The Periodic Table
Learning Objectives
By the finish of this section, you will be able to:
- State the periodic law and explain the system of elements in the periodic tabular array
- Predict the full general properties of elements based on their location within the periodic table
- Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic table
Equally early on chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together past their like chemical behaviors. One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and accept similar chemical properties. A 2nd grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemic properties in common. Yet, the specific properties of these 2 groupings are notably unlike from each other. For instance: Li, Na, and Thou are much more reactive than are Ca, Sr, and Ba; Li, Na, and Thou form compounds with oxygen in a ratio of 2 of their atoms to 1 oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to ane oxygen atom. Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also showroom similar backdrop to each other, but these properties are drastically different from those of any of the elements above.
Dimitri Mendeleev in Russia (1869) and Lothar Meyer in Federal republic of germany (1870) independently recognized that there was a periodic relationship among the properties of the elements known at that time. Both published tables with the elements arranged according to increasing diminutive mass. But Mendeleev went 1 footstep further than Meyer: He used his tabular array to predict the existence of elements that would have the properties similar to aluminum and silicon, but were yet unknown. The discoveries of gallium (1875) and germanium (1886) provided great support for Mendeleev'southward work. Although Mendeleev and Meyer had a long dispute over priority, Mendeleev'southward contributions to the development of the periodic tabular array are now more widely recognized (Effigy i).
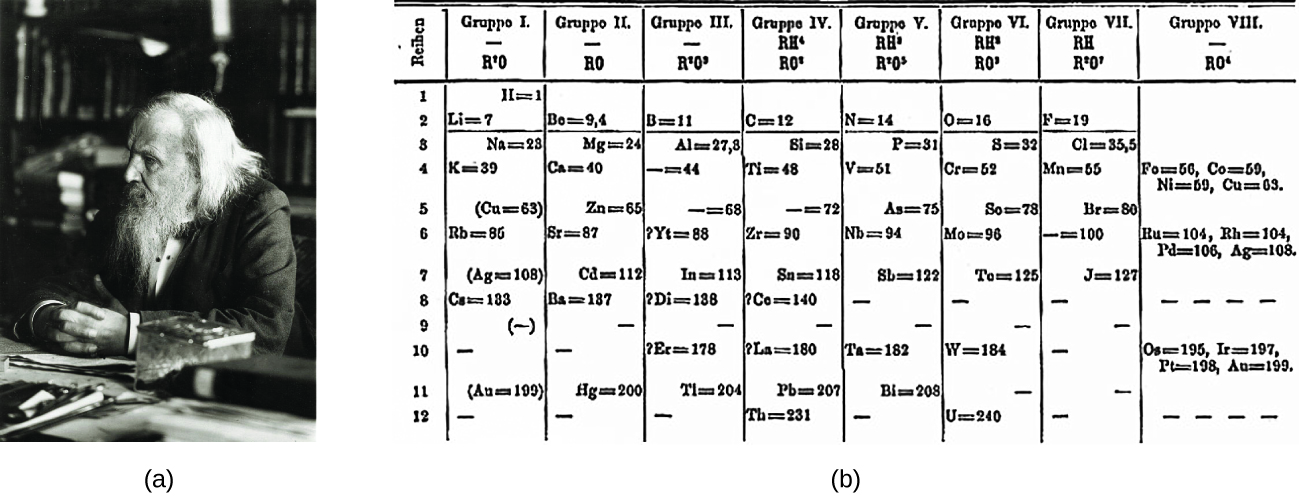
Past the twentieth century, it became credible that the periodic relationship involved atomic numbers rather than atomic masses. The modernistic statement of this relationship, the periodic law, is as follows: the backdrop of the elements are periodic functions of their atomic numbers. A modern periodic table arranges the elements in increasing society of their atomic numbers and groups atoms with similar backdrop in the same vertical cavalcade (Figure two). Each box represents an element and contains its atomic number, symbol, boilerplate atomic mass, and (sometimes) proper noun. The elements are arranged in seven horizontal rows, chosen periods or series, and xviii vertical columns, called groups. Groups are labeled at the meridian of each column. In the United States, the labels traditionally were numerals with capital letters. Still, IUPAC recommends that the numbers i through 18 be used, and these labels are more common. For the tabular array to fit on a single folio, parts of two of the rows, a full of 14 columns, are unremarkably written beneath the main body of the table.
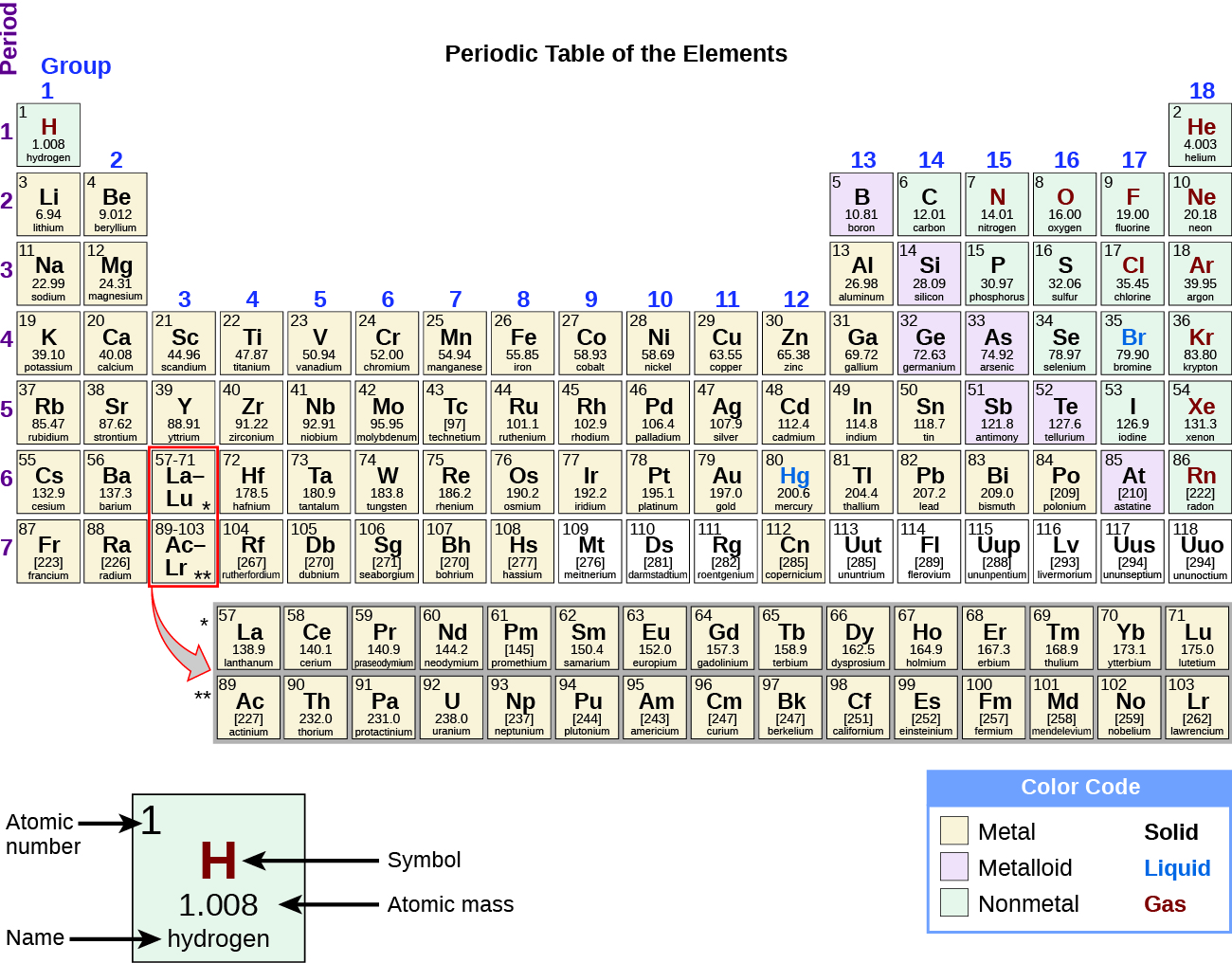
Many elements differ dramatically in their chemical and physical properties, but some elements are similar in their behaviors. For example, many elements appear shiny, are malleable (able to be deformed without breaking) and ductile (can be drawn into wires), and comport heat and electricity well. Other elements are non shiny, malleable, or ductile, and are poor conductors of heat and electricity. We can sort the elements into large classes with mutual backdrop: metals (elements that are shiny, malleable, good conductors of rut and electricity—shaded yellow); nonmetals (elements that appear wearisome, poor conductors of heat and electricity—shaded dark-green); and metalloids (elements that conduct heat and electricity moderately well, and possess some properties of metals and some properties of nonmetals—shaded royal).
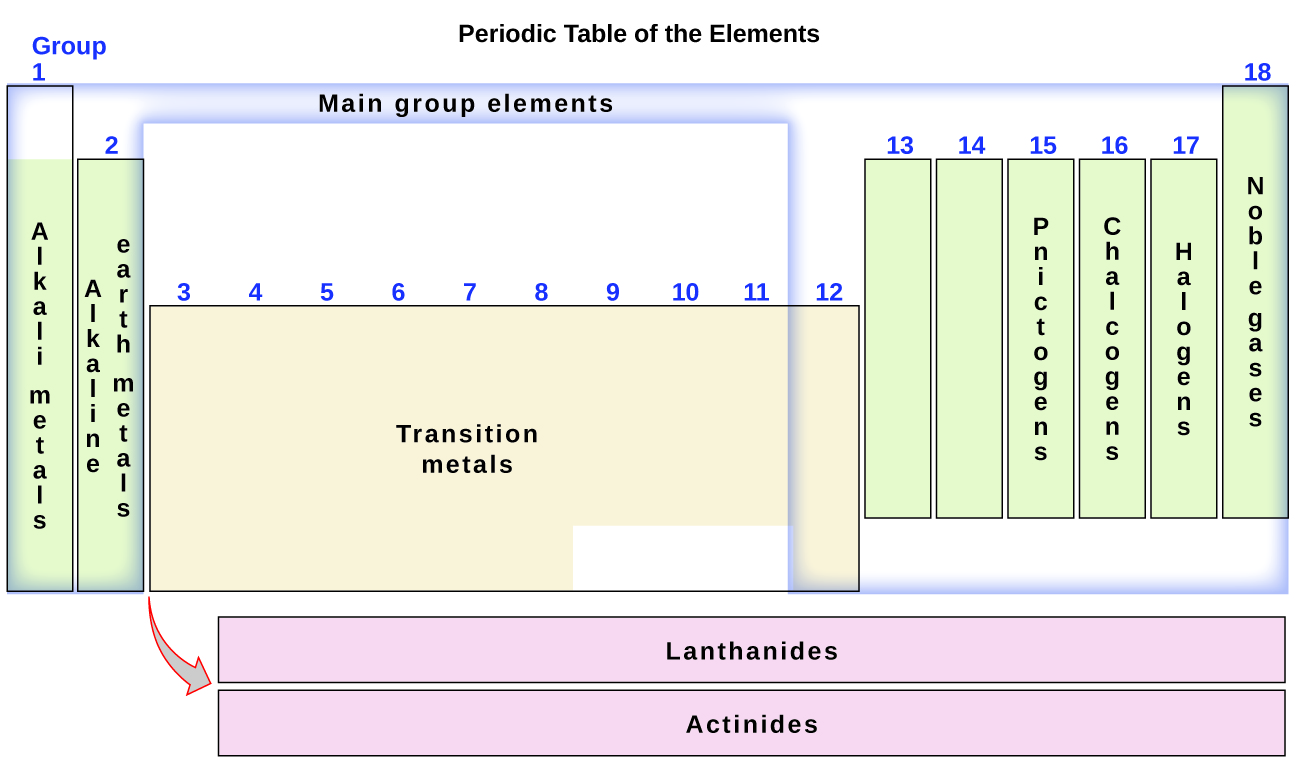
The elements can also be classified into the principal-group elements (or representative elements) in the columns labeled i, 2, and 13–xviii; the transition metals in the columns labeled iii–12; and inner transition metals in the two rows at the lesser of the table (the top-row elements are called lanthanides and the bottom-row elements are actinides; Effigy three). The elements tin can be subdivided further by more specific properties, such as the composition of the compounds they course. For example, the elements in grouping i (the first column) grade compounds that consist of 1 atom of the element and ane atom of hydrogen. These elements (except hydrogen) are known every bit alkali metals, and they all have similar chemic properties. The elements in group ii (the 2d column) course compounds consisting of 1 cantlet of the element and ii atoms of hydrogen: These are called alkaline metal earth metals, with similar properties among members of that group. Other groups with specific names are the pnictogens (grouping 15), chalcogens (grouping 16), halogens (group 17), and the noble gases (group 18, also known as inert gases). The groups can also be referred to by the outset element of the group: For example, the chalcogens can be called the oxygen group or oxygen family. Hydrogen is a unique, nonmetallic element with backdrop similar to both group 1A and grouping 7A elements. For that reason, hydrogen may be shown at the top of both groups, or past itself.

Click on this link for an interactive periodic table, which you can utilize to explore the properties of the elements (includes podcasts and videos of each element). Yous may too desire to attempt this 1 that shows photos of all the elements.
Instance 1
Naming Groups of Elements
Atoms of each of the post-obit elements are essential for life. Give the group proper name for the following elements:
(a) chlorine
(b) calcium
(c) sodium
(d) sulfur
Solution
The family unit names are as follows:
(a) halogen
(b) alkaline earth metal
(c) brine metal
(d) chalcogen
Cheque Your Learning
Give the grouping proper name for each of the following elements:
(a) krypton
(b) selenium
(c) barium
(d) lithium
Answer:
(a) noble gas; (b) chalcogen; (c) alkaline earth metal; (d) brine metal
In studying the periodic table, y'all might take noticed something nigh the atomic masses of some of the elements. Element 43 (technetium), element 61 (promethium), and most of the elements with atomic number 84 (polonium) and college have their diminutive mass given in foursquare brackets. This is washed for elements that consist entirely of unstable, radioactive isotopes (you will learn more most radioactivity in the nuclear chemistry affiliate). An boilerplate atomic weight cannot be determined for these elements because their radioisotopes may vary significantly in relative abundance, depending on the source, or may non fifty-fifty exist in nature. The number in square brackets is the diminutive mass number (and approximate atomic mass) of the near stable isotope of that element.
Primal Concepts and Summary
The discovery of the periodic recurrence of similar properties amongst the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known equally periods and columns known as groups. Elements in the same group of the periodic table have similar chemical properties. Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. Groups are numbered 1–18 from left to right. The elements in group one are known as the alkali metals; those in group two are the alkaline globe metals; those in 15 are the pnictogens; those in 16 are the chalcogens; those in 17 are the halogens; and those in xviii are the noble gases.
Chemistry Finish of Chapter Exercises
- Using the periodic table, classify each of the post-obit elements as a metal or a nonmetal, and then further classify each every bit a main-group (representative) element, transition metal, or inner transition metal:
(a) uranium
(b) bromine
(c) strontium
(d) neon
(e) gold
(f) americium
(g) rhodium
(h) sulfur
(i) carbon
(j) potassium
- Using the periodic tabular array, allocate each of the following elements as a metallic or a nonmetal, and so further classify each as a main-group (representative) element, transition metal, or inner transition metal:
(a) cobalt
(b) europium
(c) iodine
(d) indium
(e) lithium
(f) oxygen
(h) cadmium
(i) terbium
(j) rhenium
- Using the periodic table, identify the lightest member of each of the following groups:
(a) noble gases
(b) alkaline earth metals
(c) alkali metals
(d) chalcogens
- Using the periodic table, identify the heaviest member of each of the following groups:
(a) alkali metals
(b) chalcogens
(c) noble gases
(d) alkaline earth metals
- Apply the periodic table to give the proper name and symbol for each of the following elements:
(a) the element of group 0 in the same menstruum equally germanium
(b) the alkali metal earth metal in the same flow as selenium
(c) the element of group vii in the aforementioned period as lithium
(d) the chalcogen in the same period as cadmium
- Employ the periodic table to give the name and symbol for each of the following elements:>
(a) the element of group vii in the aforementioned flow as the brine metal with 11 protons
(b) the element of group ii in the same menstruum with the neutral noble gas with 18 electrons
(c) the element of group 0 in the same row as an isotope with thirty neutrons and 25 protons
(d) the noble gas in the same menstruum as gold
- Write a symbol for each of the following neutral isotopes. Include the atomic number and mass number for each.
(a) the brine metallic with 11 protons and a mass number of 23
(b) the noble gas chemical element with 75 neutrons in its nucleus and 54 electrons in the neutral cantlet
(c) the isotope with 33 protons and 40 neutrons in its nucleus
(d) the alkaline metal earth metal with 88 electrons and 138 neutrons
- Write a symbol for each of the following neutral isotopes. Include the diminutive number and mass number for each.
(a) the chalcogen with a mass number of 125
(b) the element of group vii whose longest-lived isotope is radioactive
(c) the noble gas, used in lighting, with 10 electrons and 10 neutrons
(d) the lightest element of group i with three neutrons
Glossary
- actinide
- inner transition element in the bottom of the lesser two rows of the periodic tabular array
- alkali metallic
- chemical element in grouping i
- alkaline earth metal
- element in group 2
- chalcogen
- element in group xvi
- group
- vertical column of the periodic table
- halogen
- chemical element in grouping 17
- inert gas
- (likewise, noble gas) element in group 18
- inner transition metallic
- (also, lanthanide or actinide) element in the bottom two rows; if in the first row, also chosen lanthanide, or if in the second row, likewise called actinide
- lanthanide
- inner transition metallic in the top of the bottom two rows of the periodic tabular array
- main-group element
- (also, representative element) element in columns 1, 2, and 12–18
- metal
- element that is shiny, malleable, good conductor of heat and electricity
- metalloid
- chemical element that conducts heat and electricity moderately well, and possesses some properties of metals and some properties of nonmetals
- noble gas
- (also, inert gas) element in grouping 18
- nonmetal
- element that appears dull, poor conductor of heat and electricity
- period
- (also, serial) horizontal row of the periodic tabular array
- periodic law
- backdrop of the elements are periodic role of their atomic numbers.
- periodic table
- table of the elements that places elements with similar chemical backdrop shut together
- pnictogen
- element in group xv
- representative element
- (also, main-group chemical element) chemical element in columns 1, two, and 12–eighteen
- series
- (also, menses) horizontal row of the period tabular array
- transition metal
- chemical element in columns 3–11
Solutions
Answers to Chemical science Stop of Chapter Exercises
1. (a) metallic, inner transition metal; (b) nonmetal, representative element; (c) metallic, representative chemical element; (d) nonmetal, representative element; (e) metal, transition metal; (f) metal, inner transition element; (g) metallic, transition metal; (h) nonmetal, representative element; (i) nonmetal, representative chemical element; (j) metal, representative element
3. (a) He; (b) Exist; (c) Li; (d) O
5. (a) krypton, Kr; (b) calcium, Ca; (c) fluorine, F; (d) tellurium, Te
7. (a) [latex]_{eleven}^{23}\text{Na}[/latex]; (b) [latex]_{54}^{129}\text{Xe}[/latex]; (c) [latex]_{33}^{73}\text{Equally}[/latex] ; (d) [latex]_{88}^{226}\text{Ra}[/latex];
Source: https://opentextbc.ca/chemistry/chapter/2-5-the-periodic-table/
Publicar un comentario for "Periodic Table Families Periodic Table That Shows Solids Liquids and Gases"